Metalloproteins
![]()
Although many tend to think of life as dependent on "organic", carbon-based chemistry, metals and other "inorganic" components also play key roles in living systems. Over the years, the Cramer group has studied enzymes based on Mo, Cu, Zn, W, V, Cr, and Mn. Our current interests center around Fe-S sites or Fe-S, MoFeS, and NiFeS clusters in the enzymes nitrogenase (N2ase), hydrogenase (H2ase), and CO dehydrogenase acetyl-CoA synthase(CODH/ACS). We are interested in the resting state molecular and electronic structures, the biosynthetic pathways, and the nature of the catalytic intermediates.
We are using different spectroscopic probes or toys to study catalytic sites. Our main interest involves the use of synchrotron radiation as a powerful source of x-rays that enables a variety of techniques. These include x-ray absorption fine structure (XAFS), resonant inelastic x-ray scattering (RIXS), x-ray magnetic circular dichroism (XMCD), nuclear resonant inelastic scattering (NRVS), and inelastic x-ray scattering (IXS). We also have strong campus-based spectroscopy program, including multiple FT-IR, Raman, and Mossbauer spectrometers, combined with stopped-flow and/or photolysis capabilities.
Nitrogenase (N2ase)
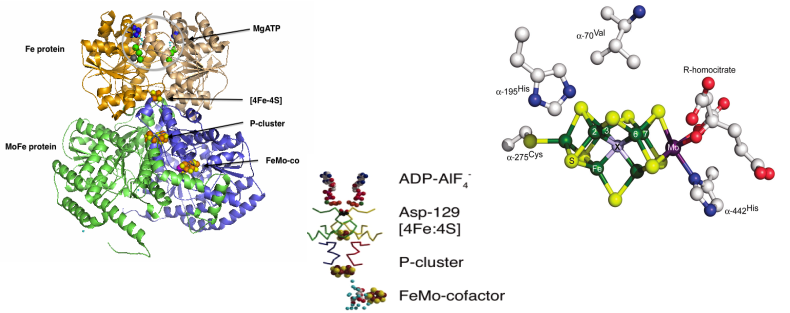
Biological nitrogen fixation involves the reduction of dinitrogen to ammonia. It is the key reaction in the nitrogen cycle and the ultimate source for most nitrogen in living systems, via the reaction:
In Azotobacter vinelandii (Av) the Mo-dependent N2ase that accomplishes this reaction uses electrons from an Fe4S4 cluster in a 63 kDa Fe protein (Av2) to reduce a 230 kDa α2β2 MoFe protein (Av1). Within Av1, an Fe8S7 ‘P-cluster’ supplies electrons to the active site MoFe7S9 ‘FeMo-cofactor’. There also exist alternative N2-reducing enzyme complexes based on VFe or even FeFe-cofactors.
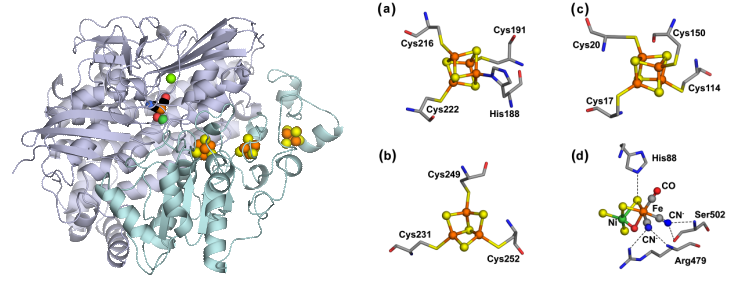
In living organisms, H2 uptake and evolution is accomplished by the enzyme H2ase:
There are three phylogenetically unrelated H2ases : (NiFe), (FeFe), and (Fe) H2ases. Apart from their biochemical significance, these enzymes have generated intense technological interest because of their relevance for a future H2 economy.
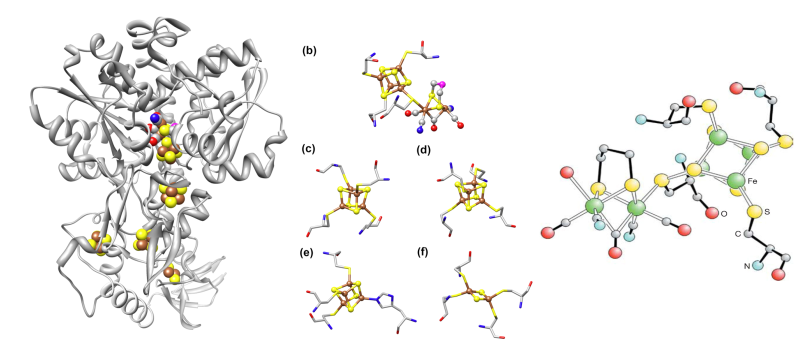
(FeFe) H2ases are among the most efficient H2 catalysts known, with Kcat values that range up to 6000 molecules of H2. [FeFe]-hydrogenase (FeFe-ase) is a prime candidate for an alternative hydrogen energy producer. FeFe-ase catalyzes the inter-conversion of H2 into electrons and protons. The active site contains a [4Fe4S] and a [2Fe2S] cluster at the catalytic site, rendering it intolerant to oxygen. There are two ways to circumvent the oxygen sensitivity of FeFe-ase; one is to provide an oxygen tolerant derivative and the other is to find a reparation pathway for the oxygen-damaged enzyme. To tackle these two solutions, we can employ the use a combination of spectroscopy techniques. It is essential to understand the motions of the active sit associated with the measured vibrational modes since these motions are a fundamental part of the catalytic process as well as of the reparation process. Experimental studies on the maturation process of FeFe-ase, a process in which the catalytic site is activated, is important for reparation pathways. A comprehensive spectroscopic study of both aspects of the enzyme; maturation and catalysis can move our energy economy towards hydrogen.
CO Dehydrogenase / Acetyl CoA Synthase (CODH/ACS)
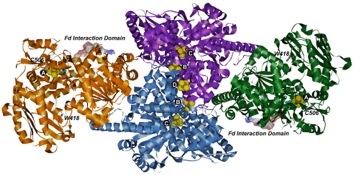
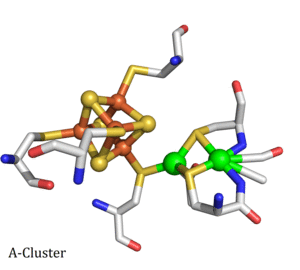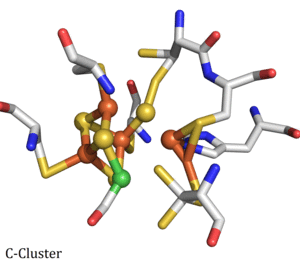
Carbon Monoxide Dehydrogenase (CODH)/Acetyl-CoA Synthase (ACS) is a double dimeric enzyme responsible for the acetylation of coenzyme-A, a key part of the ATP cycle. The CODH unit converts CO2 into CO at the C-cluster, a novel FeNiFe3S4 cluster. CO then travels down a channel to the ACS subunit. CO is combined with a methyl (CH3) group and coenzyme-A to form acetyl-CoA at the A-cluster, which is a Ni(II) cluster joined to a Fe4S4 cluster. The mechanism of CODH and ACS are of interest due to their application to carbon sequestration and sustainable hydrocarbon fuel and polymer production. We collaborate with Professor Stephen W. Ragsdale's lab at the University of Michigan on this project.

Our research is heavily dependent on synchrotron radiation sources. At the moment, we are users of the Advanced Light Source (ALS), SPring-8, the Advanced Photon Source (APS), SSRL, and PETRA-III.
IXS (Inelastic X-Ray Scattering)
http://www.rikenresearch.riken.jp/eng/frontline/5446

Analyzer Crystal
An array of four (horizontal rows) by three (vertical columns) pieces of crystal is installed at the top end of the 10m-long arm. The array is used as a spectoscope for dispersing the scattered radiation from the specimen into its components. Each piece of the analyzer crystal, 10cm in diameter, consists of 10,000 smaller pieces of perfect silicon single crystal, which are precisely arranged to form part of a sphere for the efficient collection of the scattered radiation.