HIV targets the T-cells, gaining entry through a
cascade of events mediated by the viral gylcoproteins gp120 and gp41. Gp120 is
a surface protein, noncovalently bound to the transmembrane protein gp41.
Together these two glycoproteins are presented in trimeric form on the viral
surface. Current chemotherapeutic intervention of HIV infection are directed
toward inhibiting enzymes that are required for viral replication, once a host
cell has been invaded However, the emergence of viral strains that are
resistant to these drugs has fueled investigations into alternative
intervention strategies. One active and promising area of research has focused
on preventing HIV entry into host cells. Therefore, fundamental and molecular
level understanding of the gp120 adhesion process is required in order to
develop novel metholds for detecting and deacitating the HIV virals.
Our group focuses on engineering nanostructures of HIV binding ligands to mimic
the Cellular membranes, and the regulation of the gp120 binding to these
artificially engineered structures by changing the geometry, local environment
and functionality. Approaches include (1) production of nanostructures of
ligands using scanning probe lithography and advanced nanofabrication
methodologies developed in Liu lab; (2) in situ and real time monitoring of
gp120 binding to the engineered structures to correlate the binding behavior
with the geometry, local environment and functionality of the nanostructures
and (3) binding of HIV viruses with the nanostructures of ligands.
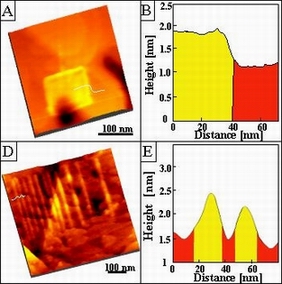
Recent progress includes: (1) successful production of nanostructures of
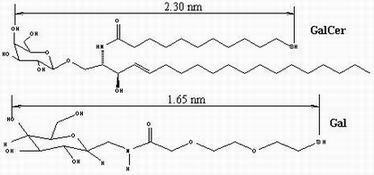
carbohydrate ligands (Gal and GalCer) using nanografting and self-assembly; (2)
immobilization of viral protein rgp120 onto Gal and GalCer terminated
self-assembled monolayers (SAMs); and (3) preliminary success in adhering
rgp120 to nanostructures of ligands.