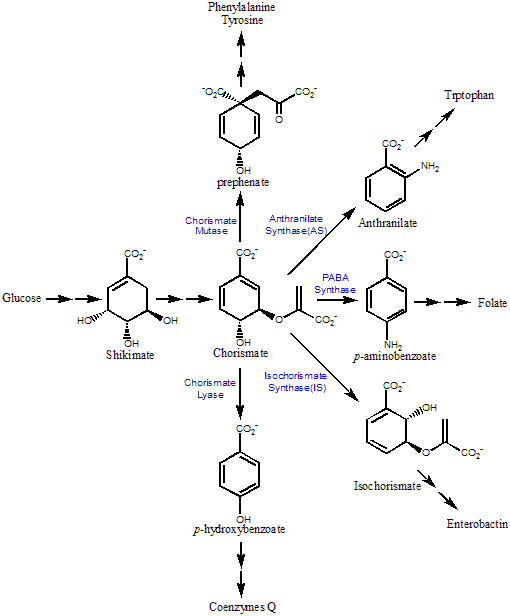
The shikimate pathway (Figure below) is found in plants, bacteria, fungi, and apicomplexan parasites. It is central to the biosyntheses of carbocyclic aromatic compounds. Chorismate is the common branchpoint for the production of these metabolites, which include aromatic amino acids, vitamins K and E, coenzyme Q, folate, enterobactin, chloramphenicol, plastoquinones, phenoxazinones, etc. Some intermediates in these pathways also have important physiological relevance. For example, salicylate is involved in plant defense against pathogens and anthranilate is the precursor of Pseudomonas quinolone signal, which regulates numerous virulence factors in Pseudomonas aeruginosa. The central biological importance of these pathways and their absence in mammals make them very attractive targets for the development of antimicrobials and herbicides. In fact, the great success of glyphosate ("Roundup") and the sulfa antibiotics, which target this pathway, has already validated this approach. We are interested in the enzymes that convert chorismate into p-aminobenzoic acid (PABA) in folate biosynthesis. Mechanistic study of the enzymes will provide critical information for the design of inhibitors, which would be lead compounds for antibiotic and herbicide development.

The first enzyme, aminodeoxychorismate
(ADC) synthase, converts chorismate and ammonia to ADC. The mechanism
is not straightforward, and still remains unresolved. The proposed
mechanism makes it all the more attractive for the design of mechanism-based
inhibitors ("suicide inhibitors"). The second enzyme,
ADC lyase, is a pyridoxal phosphate (PLP) dependent enzyme and
catalyzes the aromatization of ADC via a 1,2-elimination of pyruvate.
The chemical mechanism here is clear, whereas catalytic mechanism
and the transition state structure are not clear. The determination
of transition state structure, using proven methodologies such
as KIE analysis, can lead to the development of tight binding
transition state analog inhibitors.
end