

A wide variety of metal ions are found in the environment and in the body. They serve many functions in proteins, the most important of which is the modification of protein structures, enhancement of the structural stability of the proteins in the conformation required for biological function, or take part in the catalytic processes of enzymes. For some enzymes, the presence of metal ions is crucial for activity.


A large number of enzymes have been found to be dependent on alkali metal ions for activity. Of the alkali metal ions, as we all know, Na+ and K+ are commonly found in living systems. However, most proteins or enzymes have a catalytic preference for K+. Like K+ channel proteins, they allow K+ but not Na+ to leak through the plasma membrane. From general chemistry class, we are taught that Na+ similar to K+ except for a small difference in ion radius (0.99Å for Na+ and 1.33Å for K+). In living systems, however, this little difference has a large impact. Although the mechanisms by which alkali metal ions affect enzyme activity have very broad physiological significance they are only poorly characterized.
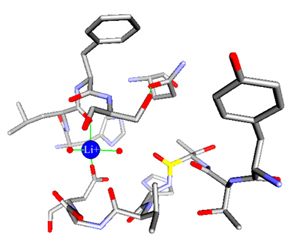

In our lab, our goal is to
study the mechanisms by which metal ions affect enzyme activity.
X-ray crystallography will be employed to illustrate their molecular
basis.
end