- Stereoelectronic
Effects
- by Emily Fogle
 Stereoelectronic
effects are simply the chemical and kinetic consequences of orbital
overlap as, for example, in the enolization of norcamphor. The
rate of the exo reaction is faster due to the developing p orbital
having better overlap with the p system and therefore the transition
state is stabilized relative to the transition state for the removal
of the endo proton. Since stereoelectronic effects were first
proposed in the 1950's, they have been demonstrated in many organic
systems and their magnitude determined both experimentally and
computationally.
Stereoelectronic
effects are simply the chemical and kinetic consequences of orbital
overlap as, for example, in the enolization of norcamphor. The
rate of the exo reaction is faster due to the developing p orbital
having better overlap with the p system and therefore the transition
state is stabilized relative to the transition state for the removal
of the endo proton. Since stereoelectronic effects were first
proposed in the 1950's, they have been demonstrated in many organic
systems and their magnitude determined both experimentally and
computationally.
Since stereoelectronic effects
play an important role in many reactions in organic chemistry,
it seems reasonable that enzymes, which have evolved to catalyze
their reactions in the most efficient manner possible, would also
employ them. There are a handful of cases where enzymes have been
demonstrated to employ stereoelectronic effects, however, their
magnitude has not been determined experimentally in any enzymatic
system.
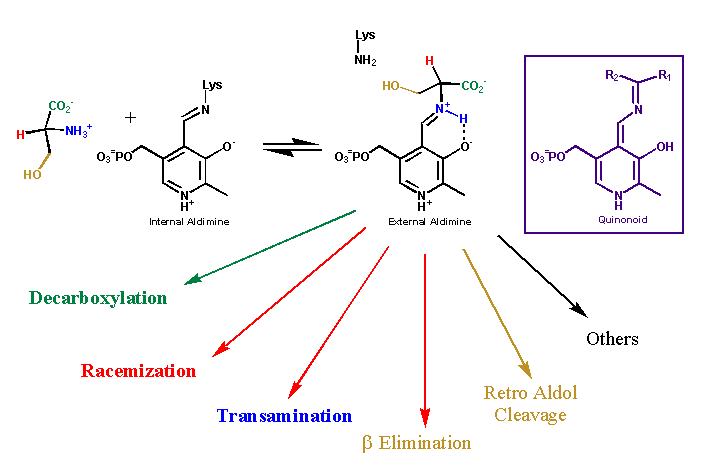
Nonenzymatic model studies
have shown that pyridoxal phosphate (PLP) is able to catalyze
transamination, racemization and decarboxylation reactions. PLP-dependent
enzymes can catalyze all these reactions and more. However, a
single PLP-dependent enzyme is quite specific, catalyzing only
a single type of reaction. This raises the question of how PLP-dependent
enzymes maintain their reaction specificity when using a cofactor
that can catalyze multiple reactions. In answer to this question,
Dunathan proposed in 1966 that PLP-dependent enzymes employ stereoelectronic
effects to control their reaction specificity.
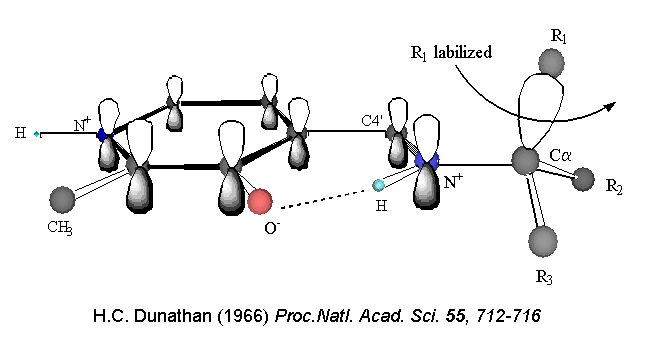
Dunathan's hypothesis held
that enzymes place the bond to be broken parallel to the p system.
In this way the developing p orbital is aligned for maximal overlap
with the extended p
system, lowering the energy of the transition state and increasing
the rate of reaction. Although Dunathan's proposal is accepted,
there is little experimental evidence to support it. One of our
main goals is to study stereoelectronic effects in PLP-dependent
enzymes in a quantitative manner.
Return
to Research Index
end
 Stereoelectronic
effects are simply the chemical and kinetic consequences of orbital
overlap as, for example, in the enolization of norcamphor. The
rate of the exo reaction is faster due to the developing p orbital
having better overlap with the p system and therefore the transition
state is stabilized relative to the transition state for the removal
of the endo proton. Since stereoelectronic effects were first
proposed in the 1950's, they have been demonstrated in many organic
systems and their magnitude determined both experimentally and
computationally.
Stereoelectronic
effects are simply the chemical and kinetic consequences of orbital
overlap as, for example, in the enolization of norcamphor. The
rate of the exo reaction is faster due to the developing p orbital
having better overlap with the p system and therefore the transition
state is stabilized relative to the transition state for the removal
of the endo proton. Since stereoelectronic effects were first
proposed in the 1950's, they have been demonstrated in many organic
systems and their magnitude determined both experimentally and
computationally.
