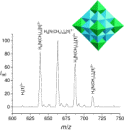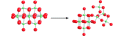
Abstract We examine oxygen-isotope exchanges in a nanometer-size oxide molecule in water and,
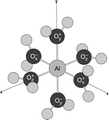
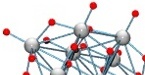
separately, both its rates of dissociation and molecular products. This molecule, the decaniobate ion
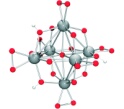
([HxNb10O28](6-x)-), is at the same size scale as geochemically
interesting features on minerals, such as surface polymers and kink sites on growth steps, although it is
structurally quite dissimilar. Unlike mineral surface structures, however, we have complete confidence in the
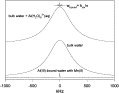
aqueous structure of this molecule and it yields a clear spectroscopic signature as it reacts. We thus can follow
proton-enhanced isotope exchanges and base-induced dissociation in unprecedented detail and clarity.
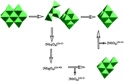
The results are surprising and require new thinking about geochemical reactions at the molecular scale. For example,
base-induced dissociation of the molecule, which is unprotonated, causes rates of oxygen-isotope exchanges of all
structural oxygens to accelerate dramatically. Similarly, protonation of the molecule causes sets of oxygens to
react, although protonation is limited. In general, all reactions are via concerted motions of many atoms and the
reactivities vary as though the entire structure was responding to changes in solution composition. The site
reactivities could not be inferred from the stable structure of the decaniobate molecule because so much of the
structure is involved in each exchange event. Thus, computational models must be structurally faithful to an
extraordinary degree, and inherently dynamic, or they will miss the essential chemistry.
Villa,E.M., Ohlin, C. A., Balogh, E., Anderson, T. M., Nyman, M. D., Casey, W. H.
Adding reactivity to structure
- reaction dynamics in a nanometer-size oxide ion in water, Am. J. Sci.,2008,
308, 942-953.Link