Design and synthesis of topologically and theoretically interesting organic molecules
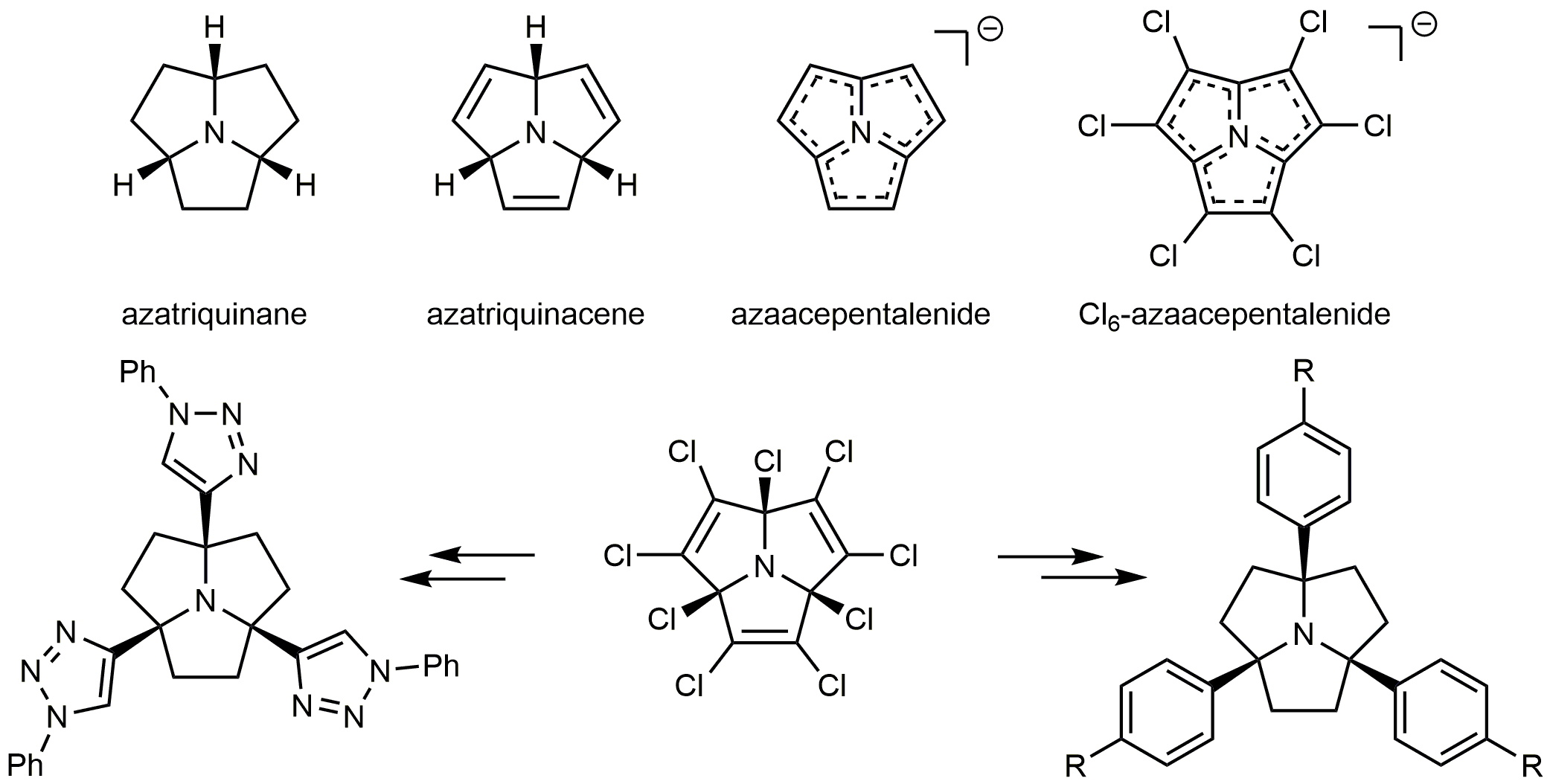
Heterotriquinanes are symmetric, convex tricycles that feature a non-invertible, apical heteroatom. The trefoil fusion of three five-membered rings results in a highly puckered structure with an acutely pyramidalized vertex. Our group was the first to complete a synthesis of azatriquinane and azatriquinacene. The bowl-shaped aromatic azaacepentalenide anion and its perchloro analogue were derived from azatriquinacene and 2,3,5,6,8,9- hexachloroazatriquinacene via the N-oxides. Nonachloroazatriquinacene can be produced in a single step and high yield by photochemical chlorination of azatriquinane. Substitution at the positions alpha to N has led both to a novel, conformationally fixed tripodal ligand system and calix-like derivatives with a basic nitrogen at the floor of the cavity.
 |
More recently, we have synthesized oxatriquinane, a fused, tricyclic alkyl oxonium ion of unprecented stability. It survives reflux in H2O, chromatography, and resists attack by alcohols, alkyl thiols, halide ions, and hindered amine bases. The X-ray crystal structure shows longer C−O bond distances and more acute C−O−C bond angles than typical alkyloxonium salts. The corresponding oxatriquinacene has also been synthesized and represents the first example of a stable allyl oxonium species. Computational modelling of oxatriquinane alongside other alkyl oxonium ions indicates that the electronic consequences of molecular strain are primarily responsible for the observed bond elongation and lack of reactivity. Chemical synthesis validated the results of the modelling work and, ultimately, an extraordinary 1.62 Å C–O bond was observed in 1,4,7-tri-tert-butyloxatriquinane. Synthesis and crystallographic analysis of the α-hydroxy oxatriquinane triflate, the first α-oxyoxonium species, revealed a 1.66 Å C−O+ bond length, which is a new record for the C−O bond.
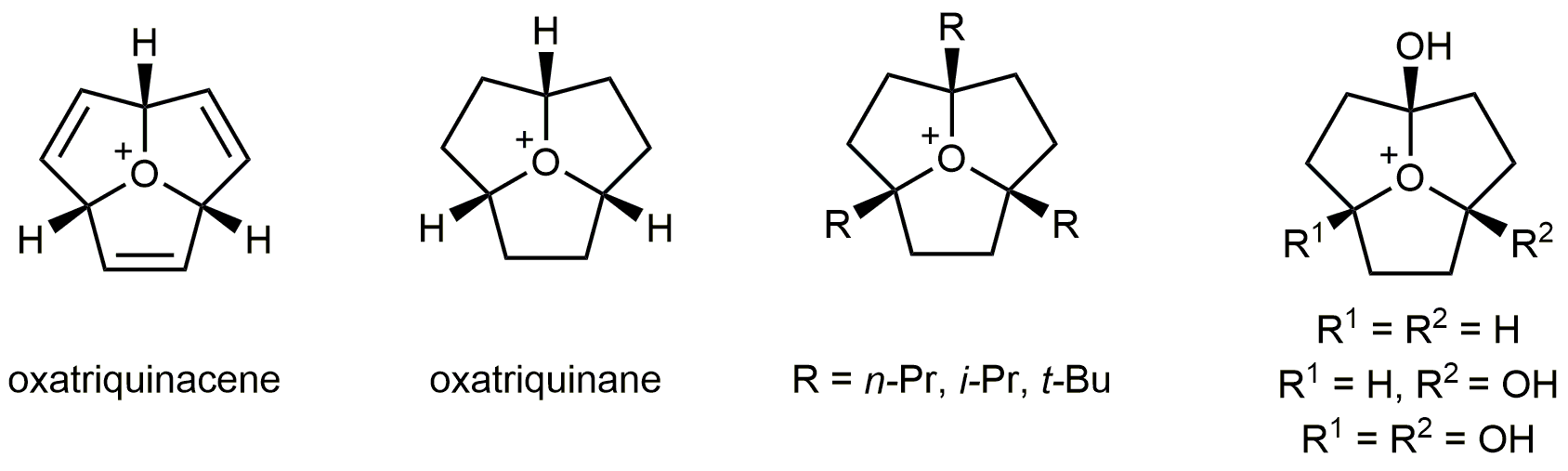 |
While trivalent oxygen species like these have been well described in the literature, the ability of oxygen to enter into a fourth covalent bonding relationship has been visited in theory and suggested by the outcome of certain reactions conducted in superacidic media, but has never been established by the characterization of a stable, persistent R3OH2+ or R4O2+ ion. We have recently shown that R3O+ oxonium cations in oxatriquinanes have experimentally verifiable basicity. In the absence of solvent, the lone pair can engage not only in conventional H-bonding with a strong acid but also in short, strong, nearly symmetrical H-bonding with a superacid such as the monomeric form of the carborane acid H(CB11Cl11). In liquid HCl, complete proton transfer from the carborane acid occurs, but the resulting R3OH2+ dication is unstable with respect to C−O bond cleavage.
Efforts in this area are currently focused on further evaluating the properties of these extraordinary molecules and the synthesis of the corresponding aromatic oxa- and thiaacepentalenes.
Lead references:
"Azatriquinanes: Synthesis, Structure, and Reactivity." N. M. Hext, J. Hansen, A. J. Blake, D. E. Hibbs, M. B. Hursthouse, O. V. Shishkin, M. Mascal, J. Org. Chem. 1998, 63, 6016.
"Azatriquinanes. Part 2. Synthesis of Azatriquinadiene and Azatriquinacene." M. Mascal, M. Lera, A. J. Blake, J. Org. Chem. 2000, 65, 7253.
"The Azatriquinenamine Trimer - a Novel Proton Chelate." M. Mascal, M. Lera, A. J. Blake, M. Czaja, A. Kozak, M. Makowski, L. Chmurzynski, Angew. Chem. Int. Ed., 2001, 40, 3696.
"Azatriquinanes. Part 4. The Chemistry of Azatriquinenamine and its Bromination Products." M. Lera, A. J. Blake, C. Wilson, M. Mascal, J. Chem. Soc. Perkin Trans. 1 2001, 3145.
"The Azaacepentalenide Anion: A New Aromatic, Bowl-Shaped Heterocycle." M. Mascal, J. Cerón Bertran, J. Am. Chem. Soc. 2005, 127, 1352.
"Ordered Crystals of Fullerenes Produced by Co-crystallization with Halogenated Azatriquinacenes." D. Pham, J. Cerón Bertran, M. M. Olmstead, M. Mascal, A. L. Balch, Crystal Growth and Design 2007, 7, 75.
"Oxatriquinane and Oxatriquinacene: Extraordinary Oxonium Ions." M. Mascal, N. Hafezi, N. K. Meher, J. C. Fettinger, J. Am. Chem. Soc. 2008, 130, 13532.
"1,4,7-Trimethyloxatriquinane: SN2 Reaction at Tertiary Carbon." M. Mascal, N. Hafezi, M. D. Toney, J. Am. Chem. Soc, 2010, 132, 10662.
"Azatriquinane as a Platform for Tripodal Metal Complexes and Calixiform Scaffolds." M. Jevric, T. Zheng, N. K. Meher, J. C. Fettinger, M. Mascal, Angewandte Chem. Int. Ed. 2011, 50, 717.
"The R3O+…H+ Hydrogen Bond: Toward a Tetracoordinate Oxadionium(2+) Ion" E. S. Stoyanov, G. Gunbas, N. Hafezi, M. Mascal, I. V. Stoyanova, F. S. Tham, and C. A. Reed, J. Am. Chem. Soc. 2012, 134, 3101.
"Extreme Oxatriquinanes and a Record C-O Bond Length" G. Gunbas, N. Hafezi, W. Sheppard, M. Olmstead, I. Stoyanova, F. Tham, M. Meyer, M. Mascal, Nature Chem. 2012, 4, 1018.
"Extreme Oxatriquinanes: Structural Characterization of α-Oxyoxonium Species with Extraordinarily Long Carbon–Oxygen Bonds" G. Gunbas, W. L. Sheppard, J. C. Fettinger, M. M. Olmstead, and Mark Mascal, J. Am. Chem. Soc. 2013, 135, 8173.
"Extraordinary Modes of Bonding Enabled by the Triquinane Framework" G. Gunbas, Mark Mascal, J. Org. Chem. 2013, 78, 9579.