Research
home · research · publications · people

Menu:
Our goal is to address challenging redox chemistry and catalysis along with applications in energy storgae and in biological systems. We are organometallic chemists, and physical inorganic chemists and we advance fundamental chemistry while studying these problems. Electricity (electron transfer reactions), coupled with chemical reactions can convert abundant molecules into a fuel. In this way, a fuel provides an energy dense, transportable means to store renewable energy. Electricity can also drive production of fine chemicals.
Group members learn organometallic synthesis and characterization, and advanced use and development of cyclic voltammetric methods to probe reaction mechanism and kinetics. Students can specialize in one or both of these areas. Examples of our current interests and projects are described below.
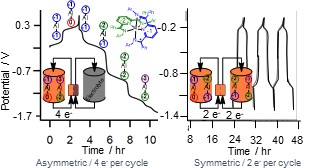
Metal Carbonyl Clusters as Models for Nanomaterials in Solar Fuels Chemistry
We are investigating multimetallic ensembles of earth-abundant iron and cobalt atoms. Soluble organometallic clusters of 4 - 13 metal atoms can be characterised precisely using the tools of molecular chemistry. Their delocalized electronic structures are most similar to those of nanomaterials and quantum dots and our studies provide insights into reaction chemistry that cannot be achieved with those less well-defined materials. We seek to control competing reactions of metal hydride catalyst intermediates such as hydrogen evolution and substrate reduction, eg. CO2 reduction to formic acid. The effects of cluster size, redox potential, tuning the secondary coordination sphere, and thermochemical vs. kinetic control of reaction pathways, are ongoing areas of investigation. Example Publications:
- Fast Proton Transfer and Hydrogen Evolution Mediated by [Co13C2(CO)24]4-
- Carr, C. R.; Taheri, A.; Berben, L. A.: J. Am. Chem. Soc. 2020, 142, 12299-12305.
- Renewable Formate from C-H Bond Formation with CO2: Using Iron Carbonyl Clusters as Electrocatalysts
- Loewen, N. D.; Neelakantan, T. V.; Berben, L. A.: Acc. Chem. Res. 2017, 50, 2362-2370.
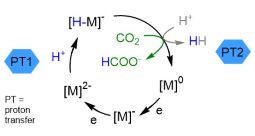
Proton-Coupled Electron Transfer Chemistry of Dihydropyridinates
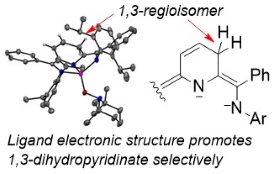
The best known dihydropyridinate is nicotinamide adenine diphosphate (NADPH) which is ubiquitous and important in the energy balance of biological systems. Generation of hydropyridinates via ligand-based proton and electron transfer events serves as a model to better understand and control the regioselectivity and chemoselectivity of hydride transfer mechanisms insynthetic systems. Organohydrides in synthetic systems have very negative redox potentials and high kinetic barriers to electron transfer. By combining pyridyl-containing ligands with Al(III) cations we lower these barriers and access milder and more selective chemistry. Example Publications:
- Ligand Conjugation Directs Formation of the 1,3-Dihydropyridinate Regioisomer
- Sherbow, T. J.; Parsons, L. W. T.; Phan, N. A.; Fettinger, J. C.; Berben, L. A.: Inorg. Chem. 2020, 59, ASAP.
- Control of Ligand pKa Values Tunes the Electrocatalytic Dihydrogen Evolution Mechanism in a Redox-Active Aluminum(III) Complex
- Sherbow, T. J.; Fettinger, J. C.; Berben, L. A.: Inorg. Chem. 2017, 56, 8651-8660.
Synthesis and Catalysis with Organometallic Group 13 Chemistry
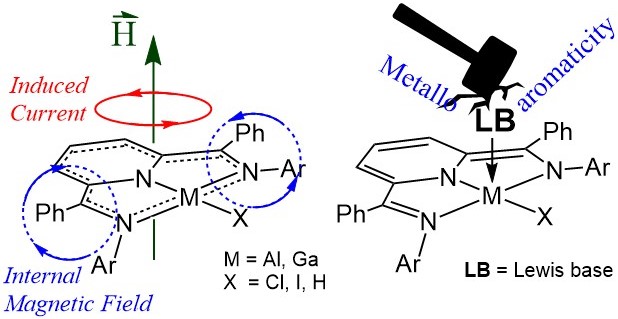
Cheap and abundant elements such as Al (8% in the earth's crust) are appealing for large scale applications such as catalysis. Explorations of square planar Group 13 complexes and their electronic structures and reactivity are areas of ongoing interest. The Berben lab accesses Al complexes in five oxidation states using redox-active ligands, and performs classic transition metal reactions such as one- or two-electron oxidation chemistry, O- or N-atom transfer, and C-H activation. Example Publications:
- Aluminum-Ligand Cooperative Bond Activation Initiates Transfer Hydrogenation Catalysis
- Carr, C. R.; Vesto, J. I.; Xing, X.; Fettinger, J. C.; Berben, L. A.: ChemCatChem 2022, 14, e20210186.
- Synthesis of Square Planar Aluminum(III) Complexes
- Thompson, E. J.; Myers, T. W.; Berben, L. A.: Angew. Chem. Intl. Ed. 2014, 53, 14132-14134.
Electron Transfer and Mixed-Valency in Organoaluminum Compounds
Our work in this area explores delocalized systems where two redox-active ligands are linked by a Group 13 ion. This fundamental chemistry has established structures that support a delocalized electronic structure. In addition to the synthesis and fundamental studies of electron delocalization we explore these compounds in applications such as nonaqueous redox-flow battery analytes that have the potential to increase energy density in grid-scale storage systems. Example Publications:
- Organic Electron Delocalization Modulated by Ligand Charge State in [L2M]n- Complexes of Group 13 Ions
- Arnold, A.; Sherbow, T. J.; Sayler, R. I.; Britt, R. D.; Thompson, E. J.; Munoz, M. T.; Fettinger, J. C.; Berben, L. A.: J. Am. Chem. Soc. 2019, 141, 15792-15803.
- A Stable Organo-Aluminum Analyte Enables Multielectron Storage for a Nonaqueous Redox Flow Battery
- Arnold, A.; Dougherty, R. J.; Carr, C. R.; Reynolds, L. C.; Fettinger, J. C.; Augustin, A.; Berben, L. A.: J. Phys. Chem. Lett. 2020, 11, 8202-8207.